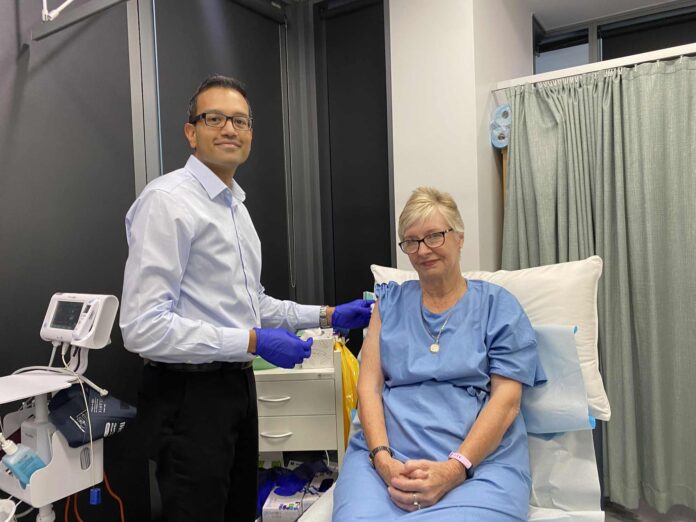
The University of the Sunshine Coast will lead trials for an investigational vaccine that could address three viruses, including COVID-19.UniSC Clinical Trials staff will enrol participants in a Moderna phase 1 trial, of a combination investigational vaccine for COVID-19, influenza and respiratory syncytial virus at its South Bank clinic in Brisbane.This will be the third time UniSC Clinical Trials conducts a trial for Moderna, to evaluate its mRNA technology.
Principal investigator Dr Nischal Sahai will oversee the trial that will assess the safety and immunogenicity of the investigational vaccine.Dr Sahai said the study presented the opportunity to evaluate the potential of investigational combination mRNA vaccines.“The investigational vaccine will be evaluated to see if it can help to reduce the burden of respiratory disease and offer the community the opportunity to be vaccinated for common respiratory viruses with one single dose and a yearly booster thereafter,” he said.“This is an investigational mRNA vaccine which contains acids that are naturally found in our body’s cells. We hope that it will instruct the body to make antibodies when exposed to these three respiratory viruses.”
The study requires healthy volunteers between the ages of 50 and 75. Participants will be asked to attend seven clinic visits and eight phone calls over a period of up to 13 months.“The investigational vaccine will be administered by injection into the upper arm muscle, like the annual influenza vaccine,” Dr Sahai said.“It is hoped that by combining the vaccines for these three respiratory viruses there could be increased protection for older people due to the ease at which it could be accessed; much like paediatric vaccines, as well as potentially increasing compliance,” he said.Eligible participants will be paid for their time on the study. Those interested in volunteering can find more information at USC – trials.
Like stories about Sunshine Coast people doing great things? Help us deliver more by registering for our free daily news feed. All it requires is your name and email. See SUBSCRIBE at the top of this article.